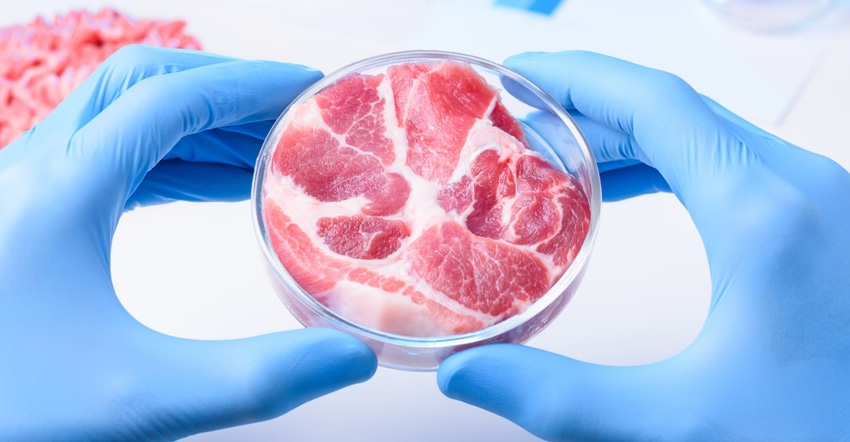
Lab meat grown from animal cells is becoming popular with each passing day, with dozens of companies making rapid strides to push the cultured meat revolution forward. According to new research, 59% of Americans have heard about lab meat. As part of the government’s oversight of the new products derived using cultured cells obtained from animals under USDA jurisdiction as they enter the marketplace, USDA’s Food Safety and Inspection Service announced an advance notice of proposed rulemaking to solicit comments and information regarding the labeling of meat and poultry products.
FSIS will use these comments to inform future regulatory requirements for the labeling of such food products.
“This ANPR is an important step forward in ensuring the appropriate labeling of meat and poultry products made using animal cell culture technology,” says USDA Deputy Under Secretary for Food Safety Sandra Eskin. “We want to hear from stakeholders and will consider their comments as we work on a proposed regulation for labeling these products.”
On March 7, 2019, USDA and FDA announced a formal agreement to jointly oversee the production of human food products made using animal cell culture technology and derived from the cells of livestock and poultry to ensure that such products brought to market are safe, unadulterated and truthfully labeled. Under the agreement, FDA will oversee cell collection, growth and differentiation of cells. FDA will transfer oversight at the cell harvest stage to FSIS. FSIS will then oversee the cell harvest, processing, packaging and labeling of products.
FDA and FSIS also agreed to develop joint principles for the labeling of products made using cell culture technology under their respective labeling jurisdictions. Seafood, other than Siluriformes fish, falls under FDA’s jurisdiction, whereas meat, including Siluriformes fish and poultry, are under FSIS’ jurisdiction.
Other than new labeling regulations concerning this product, FSIS does not intend to issue any other new food safety regulations for the cell-cultured food products under its jurisdiction, the agency said. Current FSIS regulations requiring sanitation and Hazard Analysis and Critical Control Point systems are immediately applicable and sufficient to ensure the safety of products cultured from the cells of livestock and poultry.
FSIS already has received thousands of comments on the topic in response to a 2018 joint public meeting with FDA and regarding two petitions for rulemaking (from the United States Cattlemen’s Association and Harvard Law School Animal Law and Policy Clinic). The agency, however, needs specific types of comments and information that will inform the process of developing labeling regulations for meat and poultry products made using animal cell culture technology.
The ANPR is requesting comment on specific topics to be considered during rulemaking related to statutory and regulatory requirements for the labeling of these meat and poultry products: consumer expectations about the labeling of these products, especially in light of the nutritional composition and organoleptic qualities (taste, color, odor, or texture) of the products; names for these products that would be neither false nor misleading; economic data; and any consumer research related to labeling nomenclature for products made using animal cell culture technology.
The Alliance for Meat, Poultry, and Seafood Innovation and the North American Meat Institute applaud the release. In previous comments, the groups supported regulatory processes and ultimately USDA establishing a mandatory labeling requirement for cell-based/cultured meat and poultry products.
“This is an important step in gathering information from the industry and the public to inform a labeling framework that fosters transparency, consumer confidence and a level-playing field while also aligning with longstanding law and policy, which is why we joined forces to ask the U.S. Department of Agriculture’s Food Safety and Inspection Service to take this action,” says a joint statement from NAMI and AMPS. “Both AMPS Innovation and NAMI thank USDA for their continued engagement on cell-cultured meat and poultry products and our organizations look forward to submitting comments to the agency soon.”
National Cattlemen’s Beef Association Senior Executive Director of Government Affairs Danielle Beck says NCBA is appreciative of USDA’s announcement of an advance notice of proposed rulemaking to solicit comments regarding the labeling of cell-cultured meat products.
“Cattle producers welcome competition and consumer choice, but the regulations governing alternative protein products must protect consumer health and well-being, prevent false and deceptive marketing and ensure a level playing field for real beef products,” Beck says. “We look forward to engaging in this rulemaking and will continue advocating for labeling standards which provide consumers with the appropriate information to make informed purchasing decisions.”
The U.S. Cattlemen’s Association also welcomed the announcement as an important step towards establishing a truthful and transparent labeling and regulatory framework for cell-cultured foods. As outlined in its 2018 petition for rulemaking submitted to USDA FSIS, the terms 'beef' and 'meat' should be retained exclusively for products derived from the flesh of a [bovine] animal, harvested in the traditional manner. USCA says it will be providing additional input and feedback to the requested topics in its written response.
The ANPR also discusses how FSIS will generally evaluate labels for these products if they are submitted before the agency completes rulemaking.
There is a 60-day period for comment on the ANPR. To view the ANPR and information on how to comment or submit information, visit the FSIS website.
About the Author(s)
You May Also Like






