Cellular meat production has been a topic in the livestock world throughout 2021 and it's gained quite a bit of attention. However, the real question is when it could realistically show up on grocery store shelves.
The National Coalition for Food and Agricultural Research shows some completed research by two professors and made it clear: Cellular meat production has several challenges to meet before coming to a consumer on a regular basis.
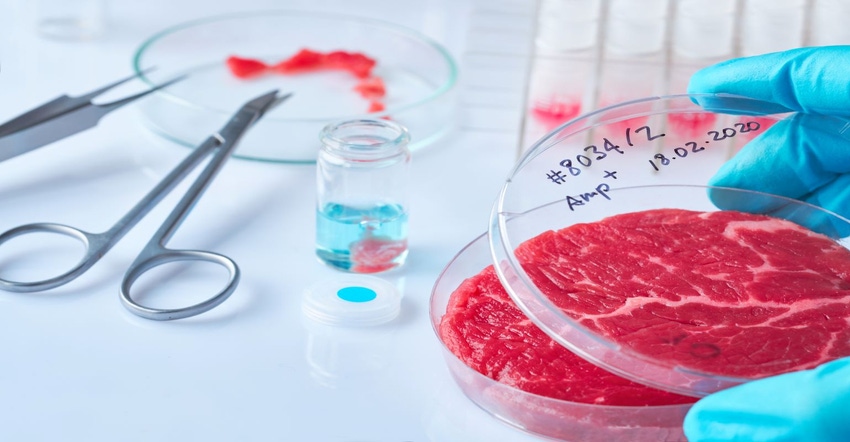
The process begins with a myoblast, or tissue, removed from cattle through a biopsy. The myosatellite cells become the foundation for cultured growth in a petri dish. These cells transform to myotubes. This, in turn, is converted to muscle fiber, or cells, which is grown around scaffolding.
Professor Keith Belk, head of animal sciences at Colorado State University, says challenges exist in the production of cellular meat. Within the department, he and assistant professor Mahesh Nair have followed such research for a while and have outlined the challenges associated with these products:
1. What to use for cell sources. Primary cells isolated from the original tissue (adult stem cells), or embryonic stem cells could be used. But this requires differentiating the meat-relevant cell types — co-culturing muscle, fat or connective tissue.
2. Right volume of cell culture media. Several growth factors must be considered for different stages. In a typical cell culture, a fetal calf, bovine or horse serum is added between 0.5% and 2% at the differentiation stage. However, volume issues can be a problem. The precise composition of the cell culture media is not defined. Several studies utilizing a serum-free media are being completed, which would use the addition of supplemental proteins (several are commercially available). However, those are not as efficient. Natural products such as mushrooms, algae and plants are being considered.
3. Antibiotics necessary. With cultured meat products, the practice is to add antibiotics or antimitotic to cells in a culture to prevent infection, particularly for long-term cultures. Patents have been filed for an antibiotic-free medium.
4. Right type of scaffolding. One issue is finding the right kind of scaffolding to get the cultured meat to grow, along with what materials should be used for the scaffolding. Should they be animal-derived or synthetic materials, like collagen scaffold or microbeads? Or should the scaffolding be made with food-grade products?
5. How to scale up production. What will be used as bioreactors? And researchers need to find a continuous supply of nutrients and growth factors. The cultured meat products will need a steady supply of oxygen in the bioreactor, and the removal of waste products id required from the cultured medium.
6. Nutritional quality. While some may tout that cellular meat production may be better for the environment, it does not mean it’s better for humans. So far, no research is available on the nutritional quality of the cell-cultured products. It doesn’t have the high levels of iron that beef has, unless the cultured meat is grown in a low oxygen environment.
7. How to assess product. Belk says due to the challenges related to the cultured meat production, cultured meat protypes are currently not available for independent technological sensorial and nutritional assessment. “It can be inferred cultured meat differs from traditional meat,” he says. “It will be at least 10 years before cultured meat will become productive.”
8. Gaining public acceptance. The jury is still out on whether cell-based products can replicate the flavor of real meat enough to be openly adopted by the general population. Belk and Nair say that more than 750 compounds are involved in beef flavor, and it is developed during cooking.
About the Author(s)
You May Also Like




