February 9, 2021

Herbicide-resistant weeds are the norm in cropping systems reliant on herbicides, with more than 500 unique cases of resistance reported worldwide in 2019. However, the ability to accumulate multiple resistances within a single population is relatively rare among weeds. Less than 10 species have biotypes resistant to five or more herbicide groups. Two Amaranthus species, waterhemp and Palmer amaranth, are among the world’s best at evolving multiple resistance. Biotypes of the two pigweeds have been identified with resistance to six different herbicide groups (HG).
Multiple-resistant biotypes are driven primarily by metabolism-based resistance. As more is learned about MBR, it is forcing researchers to rethink many assumptions regarding resistance. Target site-based resistance (TSBR) was the most common mechanism identified in early resistance research, and much of our thinking was based on these populations.
Known resistance mechanisms in waterhemp and Palmer amaranth are listed in Table 1. Dates are reported for when resistance to a herbicide group was first identified in the field, and when the resistance mechanism was reported in journals (bold date). Early research focused on TSBR, so it is unclear exactly when MBR began to appear in the Amaranthus species. For example, multiple mutations to the ALS gene were rapidly identified in many weed species, and most weed scientists simply assumed HG 2 resistance was due to these mutations.
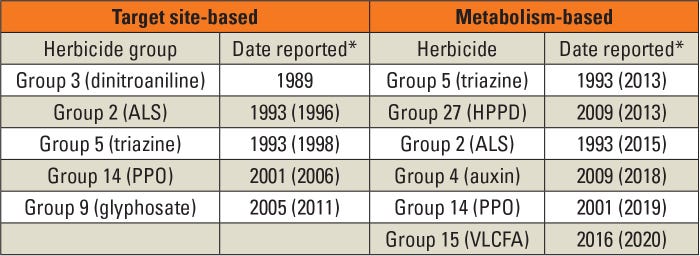
Table 1. Known resistance mechanisms in waterhemp and Palmer amaranth. *First date represents when the resistance first was reported in weed.science.org, the resistance mechanism was normally not known at this time. The date in bold and parentheses is when the resistance mechanism was first reported; most dates taken from Tranel, P. 2021. Pest Mgmt Sci. 77:43-54.
Researchers in the Jugulam Lab at Kansas State University recently identified resistance mechanisms in a six-way resistant Palmer amaranth population.
The biotype was collected from a long-term tillage study maintained in continuous sorghum for over 45 years. The population caught the attention of researchers when it survived applications of several postemergence herbicides. Herbicide options are somewhat limited in sorghum, and although a variety of herbicides were used in the research trial, 2,4-D and atrazine placed the greatest selection pressure on this Palmer amaranth population.
Five of the six resistance mechanisms identified in the Palmer amaranth population were MBR (Table 2). The only TSBR was amplification of the EPSPS gene, resulting in overexpression of EPSPS, the target site for glyphosate. This mechanism is found in other glyphosate-resistant Palmer amaranth and waterhemp populations.
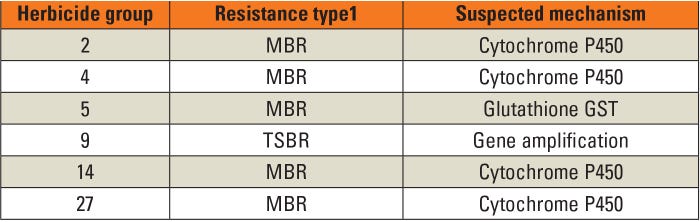
Table 2. Resistance mechanisms in a Kansas Palmer amaranth population. Source: Shyam et al. 2021. 1MBR = Metabolism-based resistance; TSBR = Target site-based resistance
There are two major enzyme families involved in metabolism of herbicides, cytochrome P450 and glutathione S-transference (GST). These enzymes provide selectivity to many of the herbicides used in crops. For example, GST is responsible for tolerance of corn to HG 5 and 15, whereas P450 is involved in metabolism of HG 2, 4 and 27. The safeners included in some herbicide formulations increase the activity of these enzymes, therefore improving crop tolerance. In this research, inhibitors of GST and P450 enzymes were used to determine the role these enzymes played in the observed resistances. GST was identified as contributing to resistance to HG 5, whereas P450 was involved with HG 2, 4, 14 and 27.
Plants have multiple versions of these enzymes, with different versions metabolizing different compounds. The specific versions of the enzymes vary among plant species. It is possible this Palmer amaranth population produces different versions of P450 or GST than susceptible Palmer amaranth populations (wild type). Another possibility is different regulation of the enzymes between resistant and susceptible populations. In this case, resistant populations would be able to increase the activity of a particular enzyme when exposed to the herbicide, whereas susceptible plants would not. Our understanding of the genetics and mechanics of MBR is limited.
The threat associated with MBR is that a single resistance mechanism (e.g., altered P450 activity) can provide resistance to multiple herbicide groups. This Kansas population possessed MBR for HG 2, 4, 14 and 27, even though most of these groups had not been used in patterns likely to select resistant biotypes. While it is possible resistant biotypes outside of the field trial introduced some of the resistances to this population via gene flow, cross-resistance to unrelated herbicide groups is well documented with MBR.
This research clearly demonstrates the threat MBR poses to our current production system. While efficient use of herbicides (e.g. multiple, effective herbicide groups) can slow the evolution of resistance, cross-resistance associated with MBR greatly reduces the effectiveness of this strategy. Minimizing the weed seed bank and adopting alternative management strategies is essential to protect the value of existing and future herbicides.
Acknowledgement: I greatly appreciate Kansas State University professor Mithila Jugulam’s willingness to review this article, and her numerous insights and explanations.
Hartzler is an Iowa State University professor of agronomy and an Extension weed specialist. KSU professor Mithila Jugulam contributed to this report.
Source: ISU Extension Integrated Crop Management, which is responsible for the information provided and is wholly owned by the source. Informa Business Media and its subsidiaries aren't responsible for any of the content contained in this information asset.
About the Author(s)
You May Also Like