by Anna Edney
Time may be running out for producers of dairy alternatives who call their beverages milk.
The Food and Drug Administration on Thursday said it’s seeking information from producers and other experts on how consumers use plant-based dairy substitutes like almond milk, the first step in what could result in a major change in how those products are labeled. The agency wants to determine whether Americans understand how the substitutes differ from cow milk in terms of nutritional content and how they react during cooking.
FDA Commissioner Scott Gottlieb said in a congressional hearing in April that the agency planned to explore whether to require the renaming of dairy alternatives labeled as milk, cheese or yogurt.
Sales of nondairy alternatives, especially soy and almond milk, have surged in recent years. Silk, a soy product made by Danone after its acquisition of WhiteWave, is expected to notch sales of $728 million this year, according to an estimate from Euromonitor.
The National Milk Producers Federation wrote to Gottlieb in October 2017 calling on him to enforce what’s known as a “standard of identity.” The FDA maintains standards of identity for many food products. The one for milk reads, in part: “the lacteal secretion, practically free from colostrum, obtained by the complete milking of one or more healthy cows.” Colostrum is the milk cows produce after giving birth.
The FDA had this fight over mayonnaise a few years ago when it warned the San Francisco-based company Just for calling its condiment mayo even though it didn’t contain eggs. The agency eventually reached a deal with Just allowing it to keep the word “mayo” on its bottle as long as it decreased the size of its logo, which used to feature an egg, and made the fact that it is egg-free more prominent.
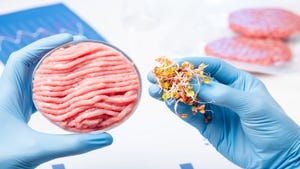
In addition to milk, the FDA is taking on lab-grown meat that relies on animal cells to produce beef, poultry and seafood. Gottlieb is holding a joint meeting with the U.S. Department of Agriculture next month to get input from the industry on the safety of the technology as well as considerations for how to label the products so consumers know they’re getting meat from a lab, not a cow.
Companies and consumers have 60 days to respond to the FDA’s questions about plant-based dairy alternatives.
--With assistance from Craig Giammona.
To contact the reporter on this story: Anna Edney in Washington at [email protected]
To contact the editors responsible for this story: Drew Armstrong at [email protected] Mark Schoifet, Timothy Annett
© 2018 Bloomberg L.P
About the Author(s)
You May Also Like